 A large number of different properties have been defined, and there are some dependencies between properties.Ī Temperature-entropy diagram ( T-s diagram) is the type of diagram most frequently used to analyze energy transfer system cycles. In general, the phases of a substance and the relationships between its properties are most commonly shown on property diagrams. Temperature-entropy Diagrams – T-s Diagrams The fact that the absolute value of specific entropy is unknown is not a problem, however, because it is the change in specific entropy (∆s) and not the absolute value that is important in practical problems. For example, the specific entropy of water or steam is given using the reference that the specific entropy of water is zero at 0.01☌ and normal atmospheric pressure, where s = 0.00 kJ/kg. Normally, the entropy of a substance is given with respect to some reference value. In general, specific entropy is a property of a substance, like pressure, temperature, and volume, but it cannot be measured directly. Because entropy tells so much about the usefulness of an amount of heat transferred in performing work, the steam tables include values of specific entropy (s = S/m) as part of the information tabulated. M = mass (kg) T-s diagram of Rankine CycleĮntropy quantifies the energy of a substance that is no longer available to perform useful work. 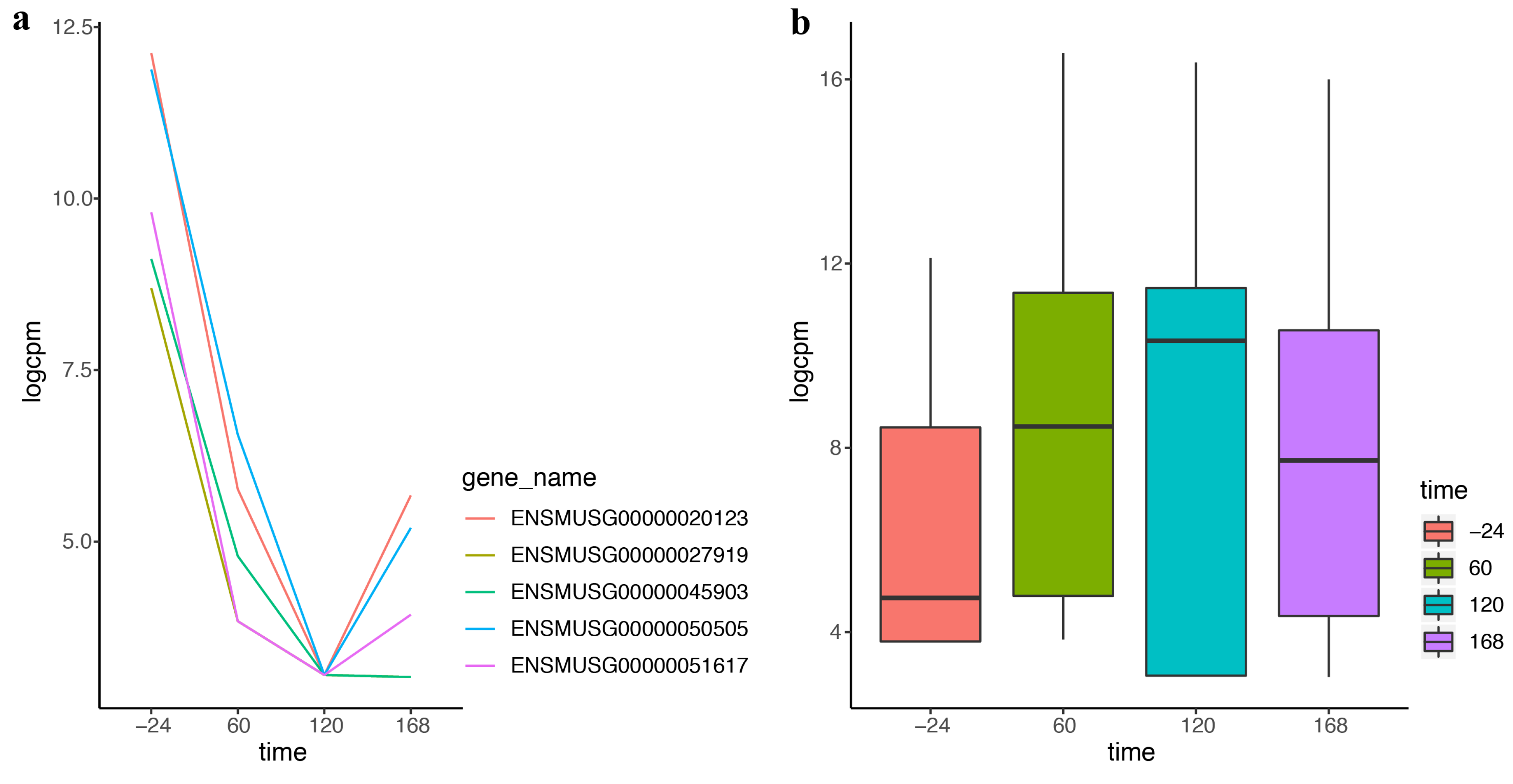 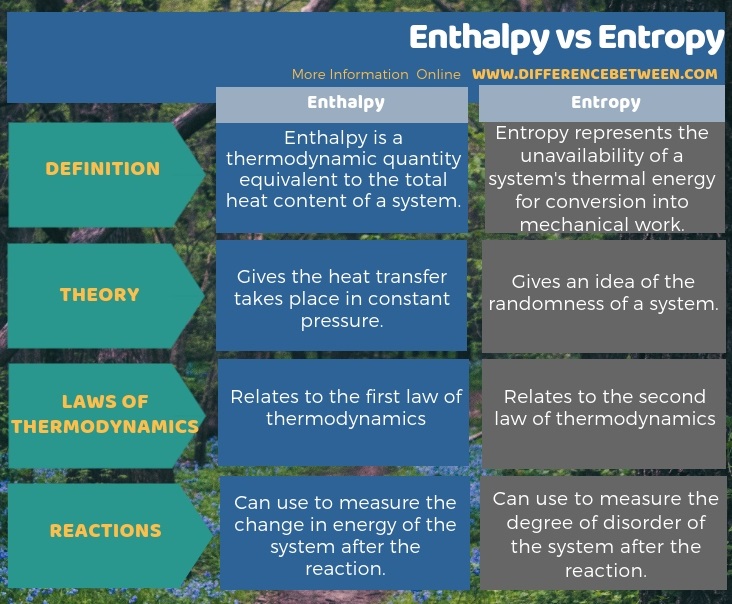
It equals to the total entropy (S) divided by the total mass (m). The specific entropy (s) of a substance is its entropy per unit mass. Engineers use the specific entropy in thermodynamic analysis more than the entropy itself. The entropy can be made into an intensive, or specific, variable by dividing by the mass. In an open system (for example, a growing tree), entropy can decrease and order can increase, but only at the expense of an increase in entropy somewhere else (e.g. In a closed system, entropy never decreases, so in the Universe entropy is irreversibly increasing. This equation, which relates the microscopic details, or microstates, of the system (via W) to its macroscopic state (via the entropy S), is the key idea of statistical mechanics. By “microscopic states”, we mean the exact states of all the molecules making up the system.Įntropy = (Boltzmann’s constant k) x logarithm of number of possible states What disorder refers to is really the number of microscopic configurations, W, that a thermodynamic system can have when in a state as specified by certain macroscopic variables ( volume, energy, pressure, and temperature). In statistical physics, entropy is a measure of the disorder of a system. Take it into your hands, shut your eyes, and twist the sides around randomly a few times.In thermodynamics and statistical physics, entropy is a quantitative measure of disorder, or of the energy in a system to do work. Imagine a 2x2 Rubik's cube, solved so that each face contains just one colour.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
